|
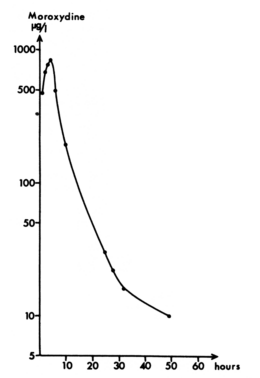
The plasma half-life of moroxydine |
Moroxydine: The Story of a Mislaid AntiviralSimon G. Sheppard BSc. Published in 1994 as Supplementum 183 to Acta Dermato-Venereologica. |
Abstract
A review is presented of the use of moroxydine in the treatment of a number of viral disorders throughout Europe and Scandinavia. The sources are unusual in their diversity and appeared in several other languages besides English. Moroxydine may be effective against a number of DNA and RNA viruses, influenza being the original application of the substance when it emerged in the late 1950s. The drug received some attention around 1960 and the discovery of the teratogenicity of thalidomide in 1961 is one likely explanation for its current obscurity. Moroxydine’s side-effects are reported to be mild and infrequent and evidence exists that the substance possesses other novel characteristics which plead for further investigation. 42 refs.
The story begins in 1957 with a patent claiming that:
The compounds especially salts of N′, N′-anhydrobis-(β-oxiethyl) biguanide, combine activity against several viruses of such different types as influenza, herpes zoster, smallpox, Newcastle disease, and canine distemper with an unusually low toxicity, resulting in a high security factor in clinical use.1
When one considers the paucity of antiviral compounds which are currently available to physicians and the evidence which is to be detailed in this review, it quite defies belief that moroxydine has not found its way into the catalogue of substances which are available for the treatment of viral disorders.
The drug originated from the work of Melander and possibly also Nilsson at the Research Department of A/B Kabi in Stockholm and the date of moroxydine’s patent now places the substance firmly into the domain of generic medicine. Moroxydine was created in an era of scientific optimism, when the popular newspapers claimed that nuclear energy would make electricity so cheap that metering would be unnecessary and scientists, it was reported, were working on a cure for the common cold. Moroxydine was probably one of those efforts since the original research was in respect of influenza. Regardless of its efficacy in any particular application however, the drug may be unique in possessing activity against a wide range of DNA and RNA viruses. One of the earliest reports of the therapeutic use of moroxydine concerned varicella zoster virus (VZV):
The new drug moroxydine was given orally or intramuscularly to 26 cases with Herpes Zoster, tablet medication is preferred since equally good therapeutic results are obtained as on parenteral administration. After two days’ adequate treatment the pains usually disappear, the temperature normalizes and propagation ceases.2
Of the 26 patients treated, 13 excellent and 8 good results were reported. This pattern is to be consistently repeated as the story of moroxydine unfolds.
1960-1965
Hardly any references to moroxydine in the mainstream, English-language medical journals have been found; one was at the end of 1960. It took the form of a rather opinionated ‘Preliminary Communication’ which appeared in the British Medical Journal. It might be surmised from the article that a good deal of publicity was being given to moroxydine around this time as an anti-influenza agent and, remarked Watson somewhat cynically, “A surprisingly large number of other virus diseases.”3 The article condemned the substance as useless but the author’s conclusions that the substance was ineffective against adenovirus and influenza infections were invalidated by the use of multiple drugs and inadequate controls. This report may have overshadowed a contemporaneous English-language account by Melander himself in a more specialized journal which provided an important summary of the early work on moroxydine.4
Only one published account was traced for 1961 and this gave a toxicological overview of ten different biguanides.5 A year later moroxydine was mentioned in another mainstream medical periodical, in this case the German journal Der Hautarzt. Almost certainly one of the underlying reasons for the obscurity of moroxydine is the fact that it was introduced at around the same time as thalidomide. Thalidomide was introduced in Britain in 1958 and withdrawn in 1961. Up to that time drug licences were granted rapidly and virtually on purity considerations alone, a situation which changed suddenly when the teratogenic effects of thalidomide became apparent. In Der Hautarzt Theodor Nasemann of the University of Munich casually remarked that “We tried moroxydine clinically in several forms of HSV infection before deciding to experiment in vitro”6 which might give an indication of the carefree attitude towards new drugs at the time. Since the original investigations with moroxydine concerned influenza, it may have been concluded in the aftermath of thalidomide that the risks of such a substance outweighed the benefits. Moroxydine might not be the only casualty in terms of promising new compounds which were abandoned following the discovery of the effects of thalidomide, although whether as fine a prize as moroxydine could ever be retreived again is uncertain.
Commenting on patients again, Nasemann went on to say that:
We did not see a definite effect in all cases but undoubtedly there was repeatedly an improvement so rapid that it could be attributed to moroxydine. In vitro, 3mg/cm³ of the substance inhibited the development of HSV infection in HeLa cells; at this concentration there were some minor signs of cellular toxicity but no cell death.
He concluded that “the substance is virustatic and not virucidal” and this remark is especially telling; the possibility that a chemotherapeutic substance might actually eradicate viral latency, rather than merely inhibit replication, is unlikely to be entertained for long today, particularly in the pessimistic atmosphere which has come to pervade. In this instance it might even be possible that Nasemann was wrong; some of the accounts which follow imply that moroxydine might be capable of inhibiting the re-establishment of latency in some cases. Some of the in vitro investigations to be related give a degree of credence to this claim.
In 1963 a Japanese patent was granted with the claim that moroxydine was useful as a remedy for diabetes mellitus.7 The nearest drug to moroxydine which is widely available at present is metformin, and this substance is effective in diabetics with some residual functioning pancreatic islet cells. The biguanide family also appears to have received attention as potential antimalarial agents. In another 1963 report moroxydine was used in the treatment of a range of viral conditions including acute meningitis and pneumonia.8 A total of 26 patients were treated, often successfully, and in the 3 cases of meningitis a rapid return to normal temperature was consistently observed. This early French trial also contains the sole account to date of the use of the drug in hepatitis.
In 1964 a series of accounts began to appear, each by different commentators, in the monthly journal Semaine Therapeutique, a supplement to Semaine des Hopitaux de Paris. The first was by Durandeau and concerned a group of patients with urethritis for whom all other treatment methods had failed:
Of 10 male patients who were carriers of viral urethritis, 8 have become cured; in 3 the laboratory has confirmed the disappearance of enclosing, the five others were not verified. The tolerance of the drug was always excellent.9
This was followed by the first report of the use of moroxydine at higher doses:
A 29-year-old man with mouth ulcers recurrent over many years was treated with various medications without success. Treatment with moroxydine at 3 × 600mg per day was given for six days and a recurrence with diminished intensity occurred after one month which was similarly treated. Over two years three outbreaks occurred much reduced in severity and duration which were easily treated with 3 × 300mg per day. It is possible that moroxydine at elevated doses offers a therapy with long-lasting results.10
Similarly, later that same year, the same pages carried the report that:
A young woman, a six-year-old infant and an aged woman were treated for manifestations of varicella zoster virus at a dose of 500mg or 900mg per day for 6–10 days. There was no recurrence in at least two of the cases and although the number of patients is small in this report, there may be a secondary action of moroxydine which merits further study.11
These accounts, written in the style of their day, undoubtedly fail to be convincing when measured against the modern objective standard of the double-blind, randomized, controlled trial which has evolved over the last few decades. However this is to take each of these reports in isolation; it is argued that the cumulation of them is otherwise and the independent accounts from so many different sources of the successful application of moroxydine are repeated so often that its usefulness seems to be beyond question. In any event, there can be no doubt about the general safety of moroxydine following early large-scale trials using the substance in the prevention and treatment of influenza. A Norwegian review documented a collection of trials involving 1,780, 596, 1,545, 4,358, 1,542 and 427 subjects with favourable, if inconsistent, results being reported.12 This account, which appeared in 1965, is the first in the present survey to use the term ‘double blind’ and the concerns which were the legacy of thalidomide are also readily apparent. The trials took place during epidemics of influenza and non-bacterial respiratory tract infections among military and industrial personnel at doses of up to 1,500mg per day. Moroxydine is evidently considered to be safe by the authorities in France and Belgium where it is available from pharmacies as an OTC preparation.
Table 1. Quoted applications of Moroxydine HCl (ABOB)
| Quoted applications | References |
| Adenovirus | 3, 12, 25, 28, 36 |
| Dengue Fever Virus | 25 |
| Hepatitis | 8 |
| Herpes Simplex Virus | 6, 8, 14, 16, 17, 18, 20, 21, 25, 27, 30, 34, 41, 42 |
| Influenza Virus | 3, 4, 12, 19, 40 |
| Molluscum Contagiosum Virus | 16, 32 |
| Papillomavirus | 39 |
| Paramyxoviruses | 13, 25, 40 |
| Pityriasis Rosea | 14, 16, 38 |
| Varicella Zoster Virus | 2, 8, 11, 14, 15, 16, 17, 18, 21, 24, 25, 30, 35 |
| Viral Meningitis | 8, 26 |
| Viral Pneumonia | 8 |
1966-1970
The next report from Semaine Therapeutique is an account of an apparently successful application of moroxydine against mumps virus:
The preventive administration of moroxydine in a creche group of 18 infants of around 2 years old at risk of contracting mumps achieved more than 83% protection from infection. This level could probably be improved if treatment was started earlier and for a longer duration, continued for a week after the expiration of the probable period of incubation. The total inoffensiveness of moroxydine authorizes a recommendation for its large scale use for the prevention of epidemics in creches and, more generally, for groups of children and infants.13
The general safety of moroxydine has evidently been apparent to the worker in this study and was subsequently objectively confirmed in the following account by Privat, again in Therapeutique. This study involved 8 cases of pityriasis rosea and a further 12 patients with different herpes-like manifestations. It ended with the conclusion:
The detailed analysis of the results obtained by this therapy reveals 17 satisfactory results and 3 doubtful. Pending clarification, we report the results of a preliminary study and will later announce our definitive conclusions.14
No trace of a subsequent report by Privat could be found.
In the meantime, we are able to give a general impression which has progressively developed during the course of this study: it is known that certain therapeutic successes are, without any doubt, the result of the use of high doses of moroxydine. The excellent clinical tolerance of this medicament does not appear to be dependent on the dose and various blood, liver and renal examinations which we regularly practice with our patients have never revealed any perturbations.
Further evidence of what is perhaps moroxydine’s particular activity against VZV is provided by another French account, this time by Chevalier and Medioni:
The sudden arrival of a child with varicella in a children’s home was followed by two simultaneous cases. A treatment of 600–900mg per day for 10 days of moroxydine, 20–27mg/Kg/day, was instituted among the 30 children, 4–15 years old, who were presumed to be susceptible. A single case of varicella appeared 13 days later. The authors underline the excellent tolerance of the medicament at this strong dose, and the likelihood of a remarkable effect of moroxydine in stopping the epidemic.15
An addendum to this report remarked that a second, similar trial six months later confirmed the result of their first study.
The only recorded suspension of treatment with moroxydine due to side-effects was in Rivoire’s large scale but uncontrolled study, published in Lyon Medical in 1967:
The general tolerance of moroxydine is excellently demonstrated despite the elevated doses we have used. In effect, more than 100 patients have followed one or more cures of 10 days with a daily dosage of 2,000mg. We have noticed only some minor digestive trouble in the adult. We note, however, in 4 children struck by varicella, we have interrupted the treatment after the appearance of hepato-intestinal trouble. Moroxydine plays a valuable role in the treatment of certain dermatological affections due to herpes, zoster, aphtes and pityriasis rose de Gibert. The action of moroxydine is more rapid, more strong and more lasting when using a dose of 2,000mg/day and in the same way its tolerance is generally excellent.16
The highest ever recorded dosage of 5,600mg per day was reported in a similar trial by Ellena, this time on a smaller scale. Moroxydine was given to 38 patients and 18 good, 11 average and 9 neutral results were recorded. “The results obtained with 2,400mg are comparable with those obtained at double the dose.”17
A year later Ayih commented on the relatively low doses used in earlier studies and detailed the use of moroxydine in the treatment of HSV and VZV in Togo, West Africa. A daily adult dose of 2,000mg was quoted. The treatment, he said,
is given often and for prolonged duration at the start of a recurrence with remarkable clinical results. There is a rapid clearing of lesions while a certain weak euphoric effect of the drug has been noticed. The good general tolerance of the drug has been verified. One case of particular note may suggest that the dosage of moroxydine be individually tailored. An 18-month infant presented with a recurrent stomatitis accompanied with sores and the general phenomena of elevated fever, stomach problems and malaise. All the treatments tried still resulted in remissions of 2–4 months with relapses. At the age of 6 years the child received moroxydine at 300mg per day for 7 days in two successive cures separated by 15 days. Notwithstanding the weak dose, the remission appears total more than 18 months since treatment with moroxydine.18
The mildly euphoric effect of moroxydine mentioned in this account raises the possibility of a psychological component in moroxydine’s action. Only further, modern trials will determine whether moroxydine is to be regarded as an aspirin or penicillin for the 21st century or a mere psychological ‘pick-me-up.’ The likelihood, of course, is that it lies somewhere in between. At the very least it appears that moroxydine is capable of counteracting the viremia and malaise which often accompanies viral infection.
At the end of this decade the focus of attention on moroxydine moved from France (or, as in the latter case, a French colony) to Eastern Europe with three reports in Hungarian journals. The first commented once again on the safety of moroxydine: “Its absorption is rapid, regardless of the route of administration. Its elimination is also rapid and it does not cumulate in the organism.”19 It was further stated that “moroxydine has definite virustatic properties.” A controlled study was detailed with the result that moroxydine possessed greater effectiveness in preventing influenza than an inactivated vaccine based on several different influenza strains:
The attack-rate was 10.7% in a group of 5,036 subjects having had no prophylaxis, 5.1% in 1,419 having had an influenza vaccine and 3.9% in 737 having been subjected to moroxydine chemoprophylaxis.
The subjects is this trial were railway workers and the report also commented on a Swedish study published several years earlier involving 1,280 workers at the Volvo factories.
A 1969 account concerned primary herpetic gingivostomatitis, recurrent herpes labialis and aphtose involving 58 patients and is an early example of the topical use of moroxydine:
A daily dose of 800mg of moroxydine and painting with concentrated moroxydine solution shortens the healing time to 5 days. Local painting with moroxydine solution in recurrent herpes labialis stops the infection around the blisters within 24 hours, the process will be encompassed and completed within 2-3 days. A local treatment with moroxydine also seems to be effective in the case of aphtosis, but because of the small number of cases no definitive conclusion can yet be made.20
Table 2. Known names and preparations of Moroxydine HCl
| Name | Form | References |
| 1022 J.D. | 100mg tablet | 8 |
| Abitylguanide | 1% eye drops | 28 |
| Albaton | ||
| Biguamor | ||
| Biguan | ||
| Bioxine | ||
| Cronoformin | ||
| Diabenide | ||
| Flumadon | ||
| Flumidin/1 | 100mg tablet | 2, 4 (Note 1) |
| Flumidin/2 | 100mg tablet | 3, 12, 30, 42 |
| Flumidin/3 | 400mg tablet | 38 |
| Influcol | 10% eye drops | (Note 2) |
| Influmin | 10/20% eye drops | 29 (Note 1) |
| Morgalin | 100mg tablet | 19, 20, 21, 27, 32 (Note 1) |
| Morgaline | ||
| Spenitol | 100mg tablet | 6 |
| Virobis | 25mg tablet | 24 |
| Vironil | 100mg tablet | 25 (Note 1) |
| Virugon | 100mg tablet | 3 |
| Virusmin | ||
| Virustat/1 | 100mg tablet | 10, 11, 13, 14, 15, 16, 17, 18, 31, 33, 34, 40 |
| Virustat/2 | 400mg tablet | 41 (Note 2) |
| Note 1: Flumidin/1 and Vironil were 100mg moroxydine + 0.1mg methatropine nitrate + 0.1mg methscopolamine nitrate. Morgalin was 100mg moroxydine + 100mg ascorbic acid + 1mg methylhomatropine bromide. Influmin eye drops were a 10% (formerly 20%) concentration in a 2% methylcellulose solution. Note 2: Influcol (Poland) and Virustat/2 (France, Belgium) are currently available forms of moroxydine. | ||
1970-1974
Yet more promising indicators of moroxydine’s activity are given in the abstract from the last of this trio of Hungarian reports:
Moroxydine has been tried out in 46 patients suffering from skin diseases of viral origin. In herpes zoster the pain subsided or disappeared completely; the time of recovery was reduced. In patients with herpes simplex the subjective symptoms disappeared; the time of recovery was shorter. Either no relapses occurred, or their form was milder and they appeared later than was usual. All of the patients with Stevens-Johnson syndrome, except one, recovered. In 2 cases mild relapses occurred.21
The results of two studies of moroxydine’s in vitro activity were reported by Galabov of the Department of Virology, ISUL, Sofia and Vilagines of the Institut Pasteur, Paris. This is the first of two reports to suggest that moroxydine may possess a two-part mechanism of action in HSV-infected cell culture, in this case with moroxydine at a concentration of 500mg/ml:
Its weak action on the viral DNA synthesis and the strong inhibition it provokes on the viral development from the 10th hour after inoculation suggests that it could lead to the formation of non-functional structural proteins.22
The second, 1971 report by Galabov and Vilagines, this time in English, confirmed the earlier observations of moroxydine’s dual action. In this exciting report the authors stated that:
The compound’s effect on the synthesis of the viral DNA is very slight in comparison with its effect on the production of mature virions at the same moment – an almost 100-fold decrease (when the compound is added immediately after the virus inoculation).23
It appears from the above that moroxydine has an effect at two different phases in the viral replicative cycle, although one is 100 times weaker than the other, at least according to measurements of virus growth in primate cell culture:
Applied during the eclipse phase (2nd–4th hour), moroxydine leads to an ensuing delay of virus reproduction in the course of six hours, the difference with the control attaining approximately 2 log at the 10th hour, after which the production of mature virions gradually comes up to that of the non-treated cells (on the 20th hour: 0.5 log), i.e. the effect is reversible. The presence of moroxydine between the 4th and 10th hour does not appreciably modify the normal course of the growth curve. It may be deduced from these data that moroxydine seems to cause the formation of a certain quantity of nonfunctional early proteins, which results in the noted decrease in the synthesis of viral DNA.
This secondary action of moroxydine provides a potential explanation for the number of accounts detailed here of the long-term, curative properties of moroxydine in treating normally recurrent viral disorders. However weak or inconsistent this secondary mechanism may be it would, if confirmed, be an extremely worthwhile area for further investigation.
The commonly held notion of a constant pool of latent virus in recurrent or persistent viral infection is as unlikely a scenario as an inexhaustible desert spring; it must be fed from somewhere. Latency must be being re-established in a cyclic fashion and it is the possible interference in this process which is of particular note in this instance.
If the history of moroxydine and the manner in which it has been neglected can be described as a catalogue of errors, the following trial might be the prize of the collection, by virtue of having been so frustratingly close. The trial took place late in 1970. A sorry picture is portrayed of the inability to eradicate the varicella which had become endemic over 3 years in a Cape Town convalescent home for coloured infants. The researcher gave moroxydine at around a tenth of the correct dose yet nevertheless claimed to have followed the manufacturers’ recommendations. To compound matters, the treatment was discontinued as soon as the disease appeared:
All 23 patients in a Home for convalescent infants received 25mg moroxydine 3 times a day (75mg/day) until the disease appeared or until discharge. No side-effects were observed but the attempt to eradicate chicken pox was unsuccessful. The staff consider that the disease may have occurred in milder form, but this is not certain nor is it good enough. Estimations of the number of crops of pocks show no difference between the control and treatment months.24
It is possible to detect a certain arrogance in some of these reports (specifically, the latter and that of Watson3), as if the inclusion of a control group suddenly made the researchers’ results infallible. It may be symptomatic of the same phenomenon to dismiss a feasible result solely because the modern protocol has not been used.
The following account, also dating from 1972, concerned moroxydine in the form of Vironil, one of three examples in this survey of the use of the substance as the main constituent of a proprietary drug combination (Table 2). Presumably this was done as a marketing device, a fashion of the period to distinguish one branded product from another. In this case it appears to have backfired dramatically. A doctor wrote:
During the past two months I have made the clinical diagnosis of dengue fever in 16 patients in this practice in Suva, Fiji. In every case, the patients had sudden relief of virtually all symptoms within 12 to 48 hours of instituting treatment with the Winthrop preparation, Vironil, and apparent cure of the condition after eight to ten days’ treatment with Vironil.25
The author of this letter to the Medical Journal of Australia went on to add himself to the number of people who had been successfully treated and described, with barely concealed delight, now being able to refer to “The day I had dengue.” He also claimed good results in controlling other viruses (including two cases of mumps virus encephalitis) and said that moroxydine was “Highly effective in the virus infections mentioned above, invariably effective in these clinically diagnosed dengue cases, and free of any side-effects in all cases in my care.” This account is especially interesting because in two young dengue patients, treatment with moroxydine was interrupted due to a shortage of supply and later resumed, with entirely consistent results. However what might be becoming to the reader a familiar syndrome in the history of moroxydine rears its head again:
Vironil almost certainly was overlooked by many medical practitioners when it was first promoted by the company, due to the fact that another product of a very similar name, produced by a different pharmaceutical firm, was promoted at the same time. The latter product was of the usual ‘cold tablet’ variety giving symptomatic relief only, so that the manufacturers’ claims and the possibilities regarding the specific antivirus activity of Vironil were apparently overlooked by many doctors. Both of these preparations were promoted during winter in Australia, and with the emphasis, by the respective representatives, on their value in treating colds and influenza, hence the confusion between the two products, except in those doctors who noted the different and unusual structure of Vironil.
In 1973 a recommendation again appeared for moroxydine’s use in acute viral meningitis.26 In such urgent circumstances it would seem prudent to give rapid treatment with a substance which, according to the postulate, has a broad spectrum of antiviral activity until a precise etiology can be determined and a more specific, effective substance used.
One school of thought holds that a broad-spectrum antiviral substance cannot exist, and so a counter-argument shall better be given. In fact this argument can be reformulated as ‘We cannot do anything about a virus which mutates.’ Clearly any virus cannot be changing so extensively or it would not be distinguishable as one; stable characteristics are being exhibited. At the risk of sounding trite, viruses in general must share common properties or they would not be recognizable as such and a substance which interfered with or exploited some mechanism common to viruses would be generally effective against them. A substance fulfilling this criterion might be described not so much as a ‘magic bullet’ as a ‘magic blanket.’
The Czechoslovaks made a contribution in 1974 with a trial of 32 HSV patients aged 4–41. The paper displays a humane outlook and includes a good general summary of moroxydine. Here ‘aura’ almost certainly refers to the prodromal phase of recurrent HSV infection:
We want to explain that enlightening on the pharmacodynamic effects of biguanides, previously not well known, was not intended to be the subject of this paper. We wanted to analyse results about the suffering caused by HSV. Best results have been obtained with HSV on the face and lips. Not so good results have been obtained with inflamation of the mucous membrane of the oral cavity (stomatitis). The patients have confirmed that the best results are obtained if treatment is begun during ‘aura,’ but experience has shown that children cannot easily detect the feeling of tension at this stage of virus seeding. The medicament is licensed by the Health Office but nonetheless was difficult to obtain. We think this biguanide derivative is a big success in the field of HSV prevention. It has a high efficiency although it does not enable full elimination of the smallest seeding in the case of each treated patient. The advantages are simple dosage methods, it is non-toxic and has a low price. An important goal is to use the drug sufficiently early i.e. during ‘aura’ of recurrency.27
Table 3. Languages, if not English, of papers quoted
| Language | References |
| Czech | 27 |
| French | 8, 9, 10, 11, 13, 14, 15, 16, 17, 18, 22, 26, 31, 33, 40, 41, 42 |
| German | 6, 28, 30, 35, 36, 38 |
| Hungarian | 20, 21, 32 |
| Japanese | 7 |
| Norwegian | 12 |
| Polish | 29 |
| Romanian | 39 |
| Swedish | 2 |
1975-1980
Moroxydine has also been used in eye disease and the Bulgarian worker Galabov appears once more as the co-author of another paper, this time using moroxydine with patients and topically. The trial might be typical of the Eastern European style, of a compassionate but confused design perhaps intended to provide the most effective mixture of therapies for the majority of patients while attempting to gather useful information at the same time. The subjects were divided into 3 groups: 31 patients, the so-called placebo group, were treated with everything except moroxydine; 104 patients were treated with everything including moroxydine and only the remaining 16 patients used moroxydine alone. Still better results might have been obtained had a stronger concentration than 1% been used however:
In an epidemic of viral keratoconjunctivitis in Bulgaria in 1972, caused by adenovirus 9, we applied moroxydine in 1% solution topically in 120 of all 151 observed patients. When the treatment could be begun in the first 3–5 days of the disease, a very favourable curing effect was noted. In most of the patients occurred a significant subjective improvement. The effect was most pronounced upon the course of the keratitis stage. When moroxydine was applied early, the observed keratitis was lighter, with slight visual impairment (with a duration of more than one month in 14.4% of the patients, as opposed to 57.3% in a placebo group), with quicker epithelialization of corneal erosions and, as a result, with fewer residual corneal opacities.28
Polish practitioners have probably accumulated the most experience treating eye-disease in this way and here we return to a trial involving moroxydine alone. The following quotation is taken from one study involving 37 cases:
Prompt improvement and healing was achieved, mainly in early superficial keratitis cases. The compound appeared also very effective in keratopaties of probable virus etiology when other therapeutic methods failed. No complications or relapses were observed during the treatment.29
The German Haneke has completed two studies of moroxydine, one in 1972 and the following one in 1976 which, excepting the present study, is the most comprehensive summary of moroxydine to appear to date, with around a dozen references to moroxydine. It is more measured than the earlier French reports but again there are encouraging pointers to moroxydine’s usefulness, especially in the treatment of VZV:
Twenty patients with herpes zoster and 20 with recurrent herpes simplex were treated with moroxydine. With zoster the feeling of sickness disappears rapidly and the skin heals quickly. Taken early, the eruption of blisters in recurrent herpes simplex is mostly suppressed, or there is a rapid healing of damage to the skin. Recurrences are not prevented however moroxydine will still work with each recurrence. No side-effects at all could be observed.30
Also in 1976 the Journal Francais d’Oto-Rhino-Laryngologie carried an unusual application of moroxydine in preventing viral deafness, but regrettably the author omitted to state the dosage used:
The author reports some observations of patients presenting with recurrent deafness of viral origin and associated with slight fever, reduced sense of smell and taste and with a herpetic eruption. He insists on the value of an antiviral treatment and of cochlear support in stopping its evolution.31
The next reference, to another paper in Hungarian, is of even greater interest. Translation from Hungarian posed the greatest difficulties in the preparation of this review and it is perhaps fortunate that an English abstract was available in this case. If this and the earlier, Australian report are confirmed it would mean that molluscum contagiosum would join dengue in the group of viruses which would seem at present to be uniquely treatable by moroxydine. Treatment for molluscum contagiosum currently appears to consist of lancing the skin eruptions with iodine.
The great majority (97.7%) of 87 African patients suffering from molluscum contagiosum healed after the oral administration for several weeks of great doses of moroxydine. The frequency, clinical forms and epidemiological data of molluscum contagiosum in Black Africa are discussed.32
This practitioner, Marton, further remarked that at the ‘normal’ adult dose of 600mg/day no results were seen; only when the dosage was increased did healing begin, after between 2 and 20 days of treatment.
A French report was found that moroxydine, among a number of other substances (some of which are still regularly used) can induce perturbation of colour vision. An enquiry to the authors as to whether the disturbance attributed to moroxydine was temporary or permanent remained unanswered, but “the molecules carrying one non-ionised atom of chlorine considerably disturb colour vision, it seems that the disturbance increases with the number of atoms carried.”33
At this point attention on moroxydine swings eastward again in the first of a collection of reports from Romania. The Romanians, almost uniquely, had been making determined efforts to find a permanent treatment for HSV-induced disease for several years: “The inefficiency or limited efficiency of some treatments may be due to the capacity of HSV to induce persistent infection, overcoming the host’s immune defence mechanisms.”34 They claimed a 22.7% cure rate following repeated administration of specific immunoglobulin plus moroxydine in a controlled study involving 110 cases of recurrent mucocutaneous herpes. Even better results were claimed in a second report (Athanasiu, Predescu, Deltin et al., 1982), but their use of intramuscular gamma-globulin (IgG) in combination with moroxydine places some of their efforts outside the scope of this survey.
By this time, at the end of the Seventies, there were two more German contributions, the first of which is almost absurd. At least these sources are consistent in demonstrating the extreme lack of knowledge which exists about the drug:
A report on a 64-year-old woman with extensive herpes zoster of the trunk. The patient presented with urinary retention which stopped after 4 tablets of the chemo-therapeutic agent moroxydine. This effect of moroxydine was unknown to the manufacturer and reports on this effect in the literature were not located.35
In this case of varicella zoster neither the doctor nor the manufacturing pharmaceutical company attributed the outcome to the antiviral action of moroxydine and the doctor somewhat improbably concluded that the drug was directly affecting the bladder sphincter.
The last account of this period took the form of a letter by Mertens and the well-known virologist Eggers, but it was uninformed and inconclusive:
On one side, a marginal effect against adenovirus cannot be excluded, but on the other side not even close to enough is known which would appear to make it meaningful to use the drug in normal clinical practice, especially against influenza.36
Eggers and especially Mertens were among the authors of a handful of subsequent reviews of antiviral treatments which appeared over the next few years but nothing of significance was added about moroxydine.
After this, reports of the therapeutic use of moroxydine continued at the incredibly slow rate of one or two per year. In comparison, a search for citations of antiviral compounds in contemporary use typically reveals between 50 and 250 published studies each year. A check of three frequently used databases (EMBASE, MEDLINE and BIOSIS) during the closing stages of this study produced 93 references to moroxydine. The actual search was for moroxydine or moroxydin or moroxidine or moroxidin, discounting duplicates. A similar search for citations of acyclovir or aciclovir, which was introduced in 1981, yielded 8,125 references.
Table 4. Recorded treatment strategies using Moroxydine HCl
| Ref. (Note 1) | Daily Dose (Note 2) | Days | Remarks/topical moroxydine |
| 2 | 25–400mg | 3–7 | In intramuscular or tablet form |
| 3 | 300–600mg | ||
| 4 | 300–900mg | 7–10 | 8 weeks of medication recorded |
| 6 | 150–600mg | 7 | |
| 8 | 600–800mg | 6–24 | |
| 9 | 900mg | 10 | Repeated after 8 days cessation |
| 10 | 900–1,800mg | 5–6 | |
| 11 | 500–900mg | 6–10 | |
| 12 | 300–1,500mg | ||
| 13 | 400mg | 10 | |
| 14 | 1200–2,000mg | 5–10 | |
| 15 | 600–900mg | 10 | |
| 16 | 2000–2,400mg | 10 | |
| 17 | 2400–5,600mg | 8–28 | |
| 18 | 400–2,000mg | ||
| 19 | 600mg | 8 | |
| 20 | 800mg | 5–8 | + local solution |
| 21 | 300–600mg | 3–9 | |
| 24 | 75mg | 7–10 | |
| 25 | 600mg | 8–10 | |
| 27 | 300–600mg | 2–4 | |
| 28 | 400mg | 2–30 | + 1% solution + 1% creme |
| 29 | – | 6–14 | 10% solution only |
| 30 | 600–1,200mg | 10 | |
| 32 | 300–2,400mg | 14–42 | No effect at ‘normal’ dose |
| 34 | 600mg | 10 | + intramuscular IgG (Note 3) |
| 38 | 800–1,200mg | 8–15 | |
| 39 | 300mg | 10 | |
| 42 | 600mg | 7 | Repeated 2, 4 or 6 times |
| Note 1: Where absent the dosages are either not stated or not applicable (e.g. in vitro studies). Note 2: Taken in two or three doses. Both infant and adult dosages are included in the range. Note 3: Repeated three times at intervals of at least one month. | |||
1982 TO 1986
Details of serum levels after ingestion of 800mg of moroxydine by a human volunteer was documented in 1982. Moroxydine’s plasma half-life is around 8 hours37 and the substance was described as being “rather hydrophilic.” The earlier observation of the Hungarians19 that absorption and elimination of the drug is rapid was confirmed.
Perhaps the single most convincing study of the effectiveness of moroxydine appeared in 1984. The trial was controlled but not double-blind and yet another intruiging query remained about whether moroxydine is virucidal or virustatic. The results of a study with 123 patients suffering from pityriasis rosea irritata were detailed:
During 1981 63 patients were treated with alb. aqu. lotion and anti-histamine. During 1982 60 patients were given immediate treatment with moroxydine-HCl at doses of 400mg three times daily for adults and 400mg twice daily for schoolchildren. In the largest and worst affected group of patients, those with skin eruptions appearing on the trunk and extremities, moroxydine-HCl shortened the therapy period by 50% compared with the same group in the control year. Overall, the shortening of unavailability for work of patients treated with moroxydine was around 50% as compared with the control groups. Even though moroxydine works not virucidally but virustatically, only one relapse was observed which occurred 12 days after return to work, possibly triggered by returning to work prematurely.38
Both entries for 1985 detail unique applications of moroxydine. The first is an application of moroxydine against papillomavirus. Only 10 children with laryngeal papillomatitis were involved, so “in spite of the very good results we obtained the number of subjects studied by us are relatively small and we cannot have a final conclusion regarding this treatment.”39 As previously mentioned, there have been a handful of reviews of antiviral treatments which make passing, almost cursory references to moroxydine, acknowledging its existence but quoting little or no literature because, presumably, hardly any could be located, for reasons that are evident from Table 2. The drug was marketed using over 17 different brand names. The following excerpt is included only because it raises the possibility of activity against another paramyxovirus: “It appears to have a preventive effect in man in respect of influenza A and possibly measles.”40
Finally in 1986 there were two further reports worthy of inclusion and again they are from the French and the Romanians. Here the French seem imprecise but unequivocal and the Romanians were more specific than usual, because this is not always their habit. Firstly the French, in a review dedicated to moroxydine:
Some frequent herpes virus diseases can be treated with moroxydine hydrochloride. The author reports studies which verified its effectiveness and good level of tolerance.41
In HSV-induced eye disease again, what appears very much like good information from Romania following a study of 77 cases:
The treatment with moroxydine hydrochloride led to a reduction of 63% and 92%, respectively, of the incidence of herpes virus type 1 and 2 antigens in the conjunctival cells, coincident with the improvement of clinical symptoms or persistent recovery.42
In conclusion, it is stressed that the present review covers the most significant portion of the entire recorded therapeutic use of moroxydine in the decades since its discovery was made public. Many of the journals quoted in this study are not included in Index Medicus, although a greater number probably appear in Excerpta Medica. Records prior to 1966 were more difficult to trace, such that two important reports4,8 were only discovered in the very closing stages of the study. There is still more to find, including some early Japanese studies which can be located using the chemical name for moroxydine hydrochloride, ABOB, as the search key; the designation by WHO of the generic name of moroxydine may have been delayed. The Chemical Abstracts designation for moroxydine hydrochloride is 3160-91-6. An acetylsalicylic acid derivative, bearing the proprietary name Assur, has also been used.
For the collation of this survey the following computer databases were used: BIOSIS, CHEMABS, EMBASE, International Pharmaceutical Abstracts, Martindale Online, MEDLINE and PASCAL. The search was made especially difficult because moroxydine has appeared under a great number of proprietary names (Table 2) and thus many separate searches had to be made during the process of retrevial. All the papers referred to were ultimately obtained save for the Japanese patent.7 At no point during the study was evidence found or any suggestion made that moroxydine was acting as an immunomodulator.
Possibly the most optimistic of the reports included here are some of the early ones in Therapeutique. Contemporary practitioners will find a still more hopeful outcome vis-a-vis the activity of moroxydine readily imaginable. The reports contained in this survey carry varying degrees of authority but considering the cumulation of them and the undoubted safety of the substance, adopting moroxydine into current use would seem an obvious step.
REFERENCES
- A/B Kabi, Stockholm. (1957). Improvements in or relating to biguanides. British Patent No. 776,176.
- Schersten, B. (1959). Treatment of Herpes Zoster with N′,N′-anhydrobis- (β-oxietyl) biguanid-HCL (ABOB). Lakertidningen 6: 3563.
- Watson, G. (1960). Virugon (Flumidin): no benefit in two respiratory diseases. British Medical Journal 2: 1785.
- Melander, B. (1960). Flumidin in experimental and clinical respiratory viroses. Arzneimittelforschung 10: 319-320.
- Sterne, J. (1961). Toxicity of dimethylguanylguanidine. Congress of the International Diabetes Federation (4th, Geneva). 1: 712-716.
- Nasemann, Th. (1962). Virustatic effect of a heterocyclic biguanide on herpes simplex virus in vitro. Der Hautarzt 13: 182-185.
- Eisai Co. (1963). N′,N′-Anhydrobis(β-hydroxyethyl)biguanide orotate. Japanese Patent No. 25,395.
- Warembourg, H. & Ducloux, G. (1963). Therapeutic trial of an antiviral product, 1022 J.D., and results. Lille Medical 8: 480-483.
- Durandeau, A. (1964). Treatment of amicrobial urethritis with Virustat. Semaine des hopitaux de Paris. Supplement: Therapeutique 40: 575-576.
- Moline, R. (1965). On a case of recurrent aphthous stomatitis treated with high doses of Virustat. Semaine des hopitaux de Paris. Supplement: Therapeutique 41: 39-40.
- Burtel, M. (1965). Antihelminthic action of Virustat. Semaine des hopitaux de Paris. Supplement: Therapeutique 41: 176.
- Bovre, K. (1965). ABOB (N,N-anhydro-bis (2-oxyethyl) biguanide chloride). Flumidin. Tidsskrift for den Norske Laegeforening 85: 934-935.
- Rouille, M. (1966). Chemoprophylaxis with Virustat in an epidemic of mumps in a group of children. Semaine des hopitaux de Paris. Supplement: Therapeutique 42: 177.
- Privat, Y. (1966). Use of Virustat in high doses in certain dermatoses of viral origin. Semaine des hopitaux de Paris. Supplement: Therapeutique 42: 549-550.
- Chevallier, J. & Medioni, D. (1967). Study of the action of a virostatic agent ABOB (Virustat) in the prevention of chickenpox in children. Semaine des hopitaux de Paris. Supplement: Therapeutique 43: 264-265.
- Rivoire, M. (1967). Action of the chlorhydrate of N1, N1 anhydrobis (beta hydroxy-ethyl) biguanide at high doses. Lyon Medical 218: 1699-1705.
- Ellena, V. (1967). Clinical trials of Virustat at very high doses. La Clinique 62: 387-388.
- Ayih, R. (1968). Virustat in African pathology. A case report. Semaine des hopitaux de Paris. Supplement: Therapeutique 44: 582.
- Lubojacky, M. & Keleti, J. (1969). Chemoprophylaxis of influenza with Morgalin. Therapia Hungarica 17: 127-129.
- Sallay, K. & Gurtler, A. (1969). Virus diseases of the mouth mucosa treated with Morgalin. Orvosi Hetilap 110: 2813-2814.
- Meszqros, C. (1970). Effect of Morgalin in the treatment of viral skin diseases. Gyogyszereink 20: 207-210.
- Galabov, A & Vilagines, R. (1970). Action of ABOB (N′, N′-anhydrobis (β-hydroxyethyl) biguanide hydrochloride) on the development of herpes virus in baboon kidney cell cultures. Arch. Gesamie Virusforch. (Archives of Virology) 31: 183-190.
- Galabov, A. & Vilagines, R. (1971). Action of N′,N′-anhydrobis (β-hydroxyethyl) biguanid hydrochloride (ABOB) on the DNA synthesis of herpes simplex virus. Doklady Bolgarskoj Akademii Nauk 24: 119-121.
- Suckling, P. (1971). Virobis (N1, N1 – anhydrobis (beta-hydroxyethyl) biguanide hydrochloride) in chicken pox. South African Medical Journal 45: 1223.
- Ashton-Martin, T. F. (1972). Treatment of dengue fever with Vironil. Medical Journal of Australia 1: 1003-1004.
- Gallais, H. & Delmont, J. (1973). Acute meningitis. Corse Mediterranee Medecine 19: 137-144.
- Kleibl, K. (1974). Biguanides in the prevention of relapsing herpetic infections. Czechoslovak Dermatology 25: 623-625.
- Wassileva, PIv. & Galabov, A. S. (1975). Treatment of epidemic keratoconjunctivitis with ABOB. Klin Monatsbl Augenheilkd 166: 77-83.
- Debicka, A. (1976). Trials of treatment of corneal virus diseases by ABOB solution. Klin Oczna 46: 639-641.
- Haneke, E. (1976). On the treatment of Herpes zoster and recurrences of Herpes simplex with Moroxydin (ABOB). Therapiewoche 26: 5508-5509.
- Chavanne, L. (1976). Recurrent viral deafnesses. Considerations on the diagnosis and prognosis of viral deafness. Journal Francais d’Oto-Rhino-Laryngologie-Audiophonologie-Chirurgie Maxillo-Faciale 25: 595-650.
- Marton, K. (1976). Symtomatology and epidemiology of molluscum contagiosum in Black Africa and its effective treatment with Morgalin. Borgyogyaszati es Venerologiai Szemle 52: 262-268.
- Laroche, J. & Laroche, C. (1977). New researches on the modification of colour vision under the action of medicaments at therapeutic doses. Annales Pharmaceutiques Francaises 35: 173-179.
- Mitiu, A., Sahnazarov, N., Baba, C. et al. (1979). Some results obtained by the ‘Stefan S. Nicolau’ Institute of Virology in the study of herpes simplex virus. Revue Romaine de Medecine: Virologie 30: 295-306.
- Grimmeke, F. (1979). On spasm of the bladder sphincter in a case of herpes zoster. Geburthshilfe und Frauenheilkund 39: 209.
- Mertens, Th. & Eggers, H. J. (1980). Moroxydine (letter). Deutsche Medizinische Wochenschrift 105: 184.
- Vessman, J., Johansson, M. & Groningsson, K. (1982). Determination of moroxydine in biological fluids by electron-gas chromatography. Journal of Chromatography 229: 227-233.
- Grobe, J. W. (1984). Virustatic effect in the treatment of pityriasis rosea irritata with moroxydine hydrochloride. Dermatosen in Beruf und Umwelt 32: 25.
- Sarau, M., Topciu, V. & Marin, I. (1985). Flumidin in the treatment of laryngeal papillomatosis in children. Revista de Chirurgie, Oncologie, Radiologie, O.R.L., Oftalmologie, Stomatologie. Oto-Rino-Laringologie 30: 93-98.
- Coquin, Y. (1985). Antiviral medicines. Revue du Praticien 47: 2867-2871.
- Quivy, F. (1986). Value of moroxydine in the treatment of herpes virus disease. Comptes Rendues de Therapeutique et de Pharmacologie Clinique 4: 7-8.
- Athanasiu, P., Anghelescu, S., Deltin, L. et al. (1986). Incidence of herpetic antigens types 1 and 2 revealed by immunoflourescence in patients suffering from chronic or recurrent keratoconjuctivitis submitted to an etiological therapy. Revue Romaine de Medecine: Virologie 37: 247-252.